What is ADR?
The ADR regulations provide guidelines and requirements for the transport of dangerous goods, including clinical waste, by road. Compliance with ADR ensures the safety of both the transport personnel and the environment.
ADR covers various aspects of transportation, including packaging, labelling, marking, documentation, and driver and vehicle requirements. The regulations aim to minimise the risks associated with the transport of hazardous materials, including clinical waste.
Packaging and Container Requirements
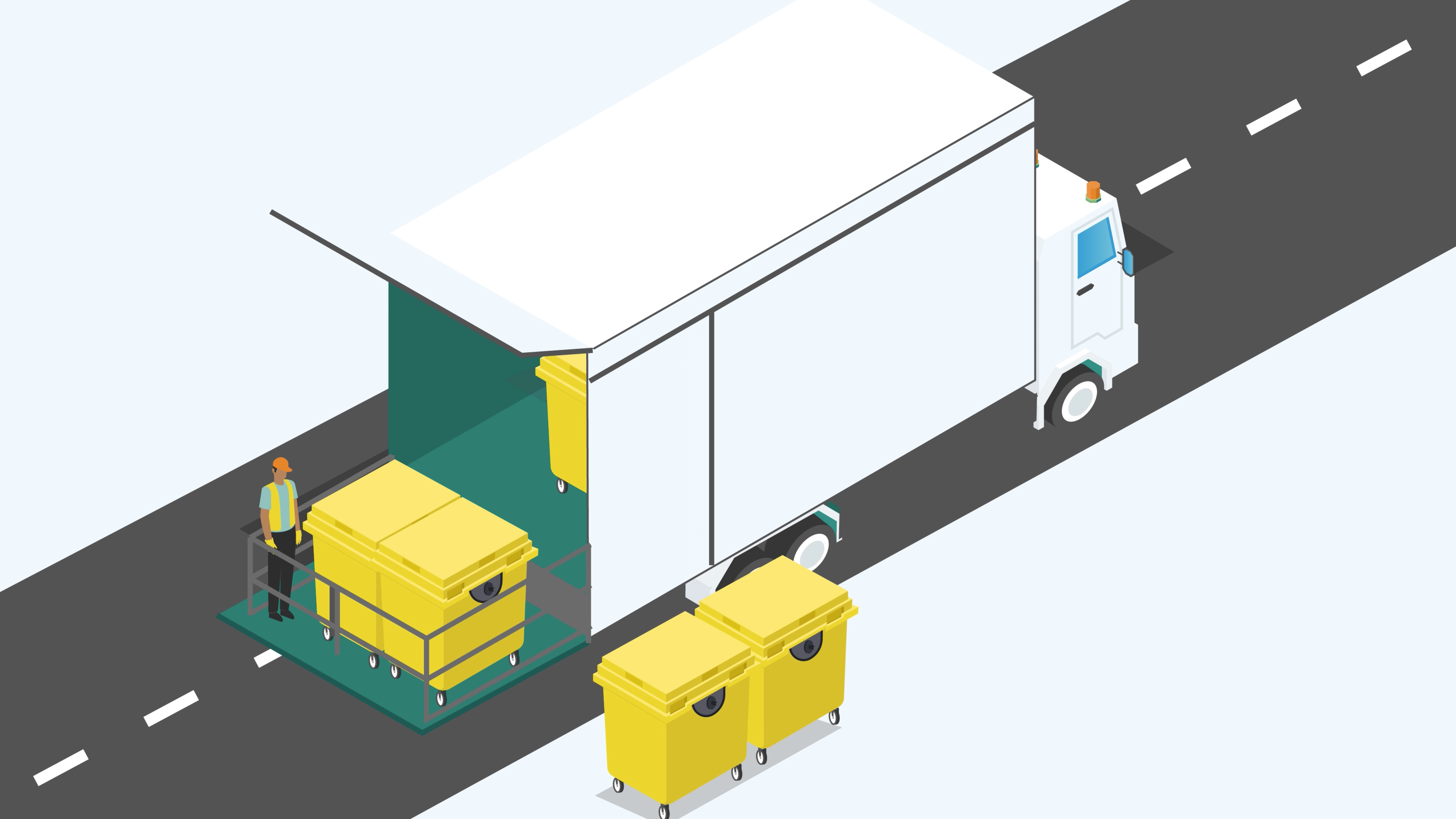
Clinical waste must be packaged and contained appropriately to prevent potential risks during transportation. The packaging and containers used for clinical waste should comply with specific standards to ensure safe transport.
Key considerations include:
- Leak-proof and Secure Containers: Clinical waste must be stored in containers that are leak-proof and securely sealed to prevent any potential release or contamination during transportation.
- Proper Labelling and Marking: All containers carrying clinical waste must be clearly labelled and marked with appropriate hazard symbols, waste type identification, and relevant information required by ADR regulations.
- Compatibility: Compatibility of the waste with the packaging materials is crucial to prevent any chemical reactions or interactions that may compromise the integrity of the containers during transportation.
- Segregation: Different types of clinical waste should be segregated appropriately within the containers to prevent cross-contamination or mixing of incompatible waste streams.